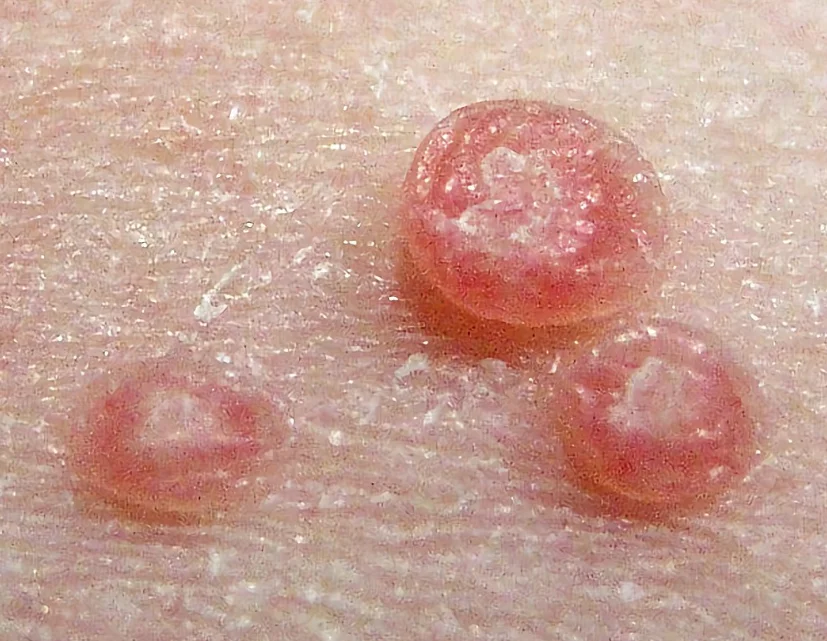
The FDA approved Berdazimer Gel 10.3% (Zelsuvmi) for Molluscum Contagiosum on 5th January 2024.
Berdazimer Gel 10.3% (Zelsuvmi) is the second topical medicine approved so far for the treatment of Molluscum Contagiosum (MC). The first one, cantharidin, was approved in 2023.
Curettage is more effective than Cantharidin. However, it has not been compared with Berdazimer topical gel.
In addition, other medicines, not approved are also commonly used to treat MC. These include keratolytic agents such as salicylic acid and vitamin A derivatives.
How does Berdazimer Gel (Zelsuvmi) heal lesions of Molluscum Contagiosum?
The primary mechanism of action of Berdazimer (Zelsuvmi) Gel is via the release of Nitric Oxide when it is applied directly over the lesions.
Nitric oxide has two different mechanisms by which it heals the lesions of MC [Ref]:
- Direct antimicrobial effects
- Immunomodulatory effects
Nitric oxide releases oxygen-free radicals at the site of the application, killing poxvirus.
In addition, nitric oxide helps fight the virus causing MC by affecting certain proteins and regulating a molecule called NF-κB.
The gel mainly acts on the skin where it’s applied, reducing the chances of side effects in the rest of the body.
How effective is Berdazimer Gel?
Berdazimer is among the two FDA-approved topical treatments of MC. However, it is more potent than Cantharidin, which was approved in 2023.
In a randomized clinical trial, Berdazimer was more effective than placebo in clearing MC lesions after 12 weeks. 32.4% (144 patients) achieved complete clearance of all MC lesions in the Berdazimer group compared with 19.7% in the placebo group [Ref]
In an analysis of 3 randomized clinical trials, Berdazimer performed better than the placebo at week 12, with a complete clearance rate of 30.0% compared to 19.8% for the placebo [Ref].
How Safe is Berdazimer Topical Gel?
Skin-related side effects are common. However, side effects are generally mild in nature.
Mild to moderate pain, erythema, and pruritus were more commonly reported with Berdazimer compared to placebo, indicating some local skin reactions associated with it.
| Read: |